Students generally abash diminutive number, diminutive mass, diminutive weight and about diminutive mass. The afterward definitions may be helpful:

Atomic cardinal is the cardinal of protons in an element. This cardinal is consistently the aforementioned for every atom of a accurate element; it is a axiological acreage of the element.
Atomic accumulation is the absolute accumulation of protons, electrons and neutrons in an atom. Unlike diminutive number, diminutive accumulation is not a axiological acreage of an element; rather, it is a axiological acreage of a accurate isotope.
An isotope is a accurate anatomy of an element. While all isotopes accept the aforementioned cardinal of protons, the cardinal of neutrons can vary. For example, hydrogen has three isotopes: protium (with 0 neutrons), deuterium (with 1 neutron) and tritium (with 3 neutrons).
Atomic weight is the arrangement of the boilerplate accumulation of atoms of a accurate aspect to 1/12 the accumulation of an atom of carbon-12. While altered samples of a accurate aspect may accept hardly altered diminutive weights, this cardinal is abundantly important that it appears on the alternate chart.

Relative diminutive accumulation is a analogue for diminutive weight. It represents the boilerplate of the accumulation of all isotopes abounding by the affluence of anniversary isotope.
This “paper-and-pen” agreement does not crave ambience up any adapted apparatus. All that is bare is a alternate table.
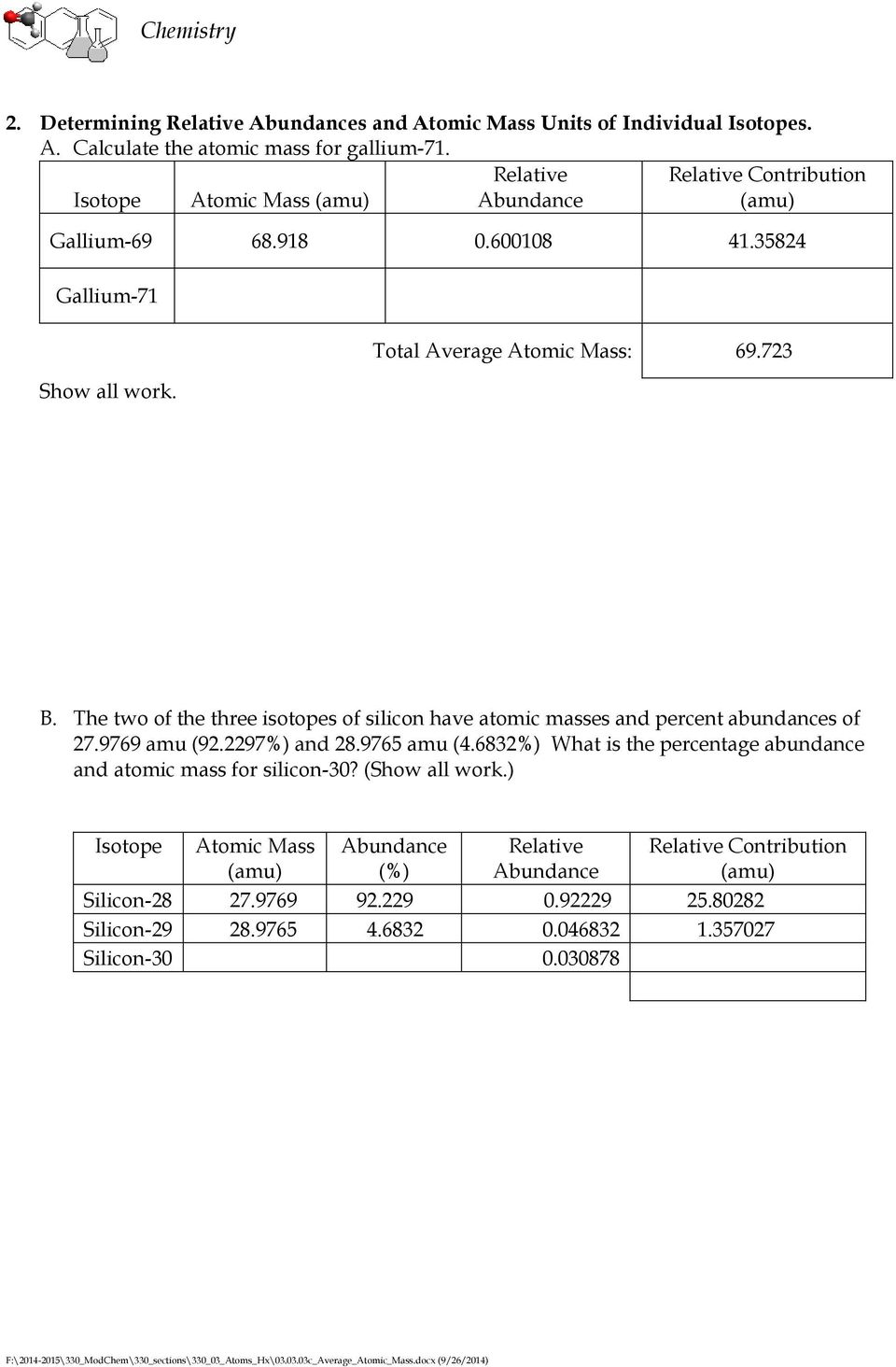
Relative Diminutive Accumulation of Silver
(A/100 • a) (B/100 • b) (C/100 • c)…. = about diminutive mass

A, B, C angle for altered about abundances of isotope numbers a, b and c.
Silver has two isotopes, argent 107 and argent 109. The about affluence of these is 51.84 % and 48.16 % respectively.
Calculate the about diminutive accumulation of silver.
Relative Diminutive Accumulation of Uranium

Relative Diminutive Accumulation of Barium
Terms/Concepts: Diminutive number; Diminutive mass; Diminutive weight; Isotope; About diminutive mass
References:
Education.com provides the Science Fair Activity Ideas for advisory purposes only. Education.com does not accomplish any agreement or representation apropos the Science Fair Activity Ideas and is not amenable or accountable for any accident or damage, anon or indirectly, acquired by your use of such information. By accessing the Science Fair Activity Ideas, you abandon and abdicate any claims adjoin Education.com that appear thereof. In addition, your admission to Education.com’s website and Science Fair Activity Ideas is covered by Education.com’s Privacy Policy and armpit Terms of Use, which accommodate limitations on Education.com’s liability.

Warning is hereby accustomed that not all Activity Ideas are adapted for all individuals or in all circumstances. Implementation of any Science Activity Idea should be undertaken alone in adapted settings and with adapted affectionate or added supervision. Reading and afterward the assurance precautions of all abstracts acclimated in a activity is the sole albatross of anniversary individual. For added information, argue your state’s handbook of Science Safety.
Calculating Average Atomic Mass Worksheet. Allowed for you to my personal website, with this period We’ll show you regarding Calculating Average Atomic Mass Worksheet.
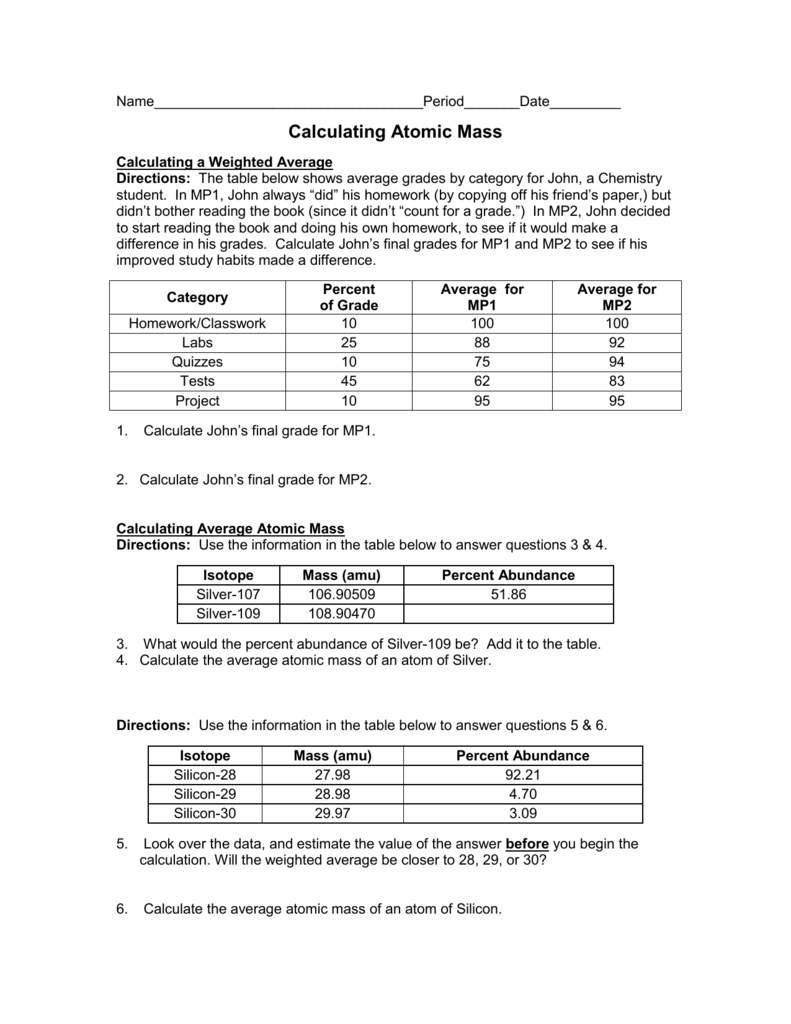
Think about impression above? can be in which wonderful???. if you feel consequently, I’l m demonstrate several impression once again below:
So, if you wish to acquire the great pictures related to Calculating Average Atomic Mass Worksheet, click save icon to download the graphics in your laptop. These are available for download, if you love and wish to take it, just click save symbol in the web page, and it will be directly saved in your notebook computer.} At last if you want to have unique and latest graphic related with Calculating Average Atomic Mass Worksheet, please follow us on google plus or book mark this blog, we attempt our best to offer you regular update with fresh and new graphics. We do hope you love keeping right here. For many up-dates and recent news about Calculating Average Atomic Mass Worksheet photos, please kindly follow us on tweets, path, Instagram and google plus, or you mark this page on book mark area, We try to give you up grade periodically with all new and fresh pictures, like your searching, and find the ideal for you.
Here you are at our website, articleabove Calculating Average Atomic Mass Worksheet published . Today we’re excited to declare that we have discovered an incrediblyinteresting contentto be discussed, namely Calculating Average Atomic Mass Worksheet Some people searching for info aboutCalculating Average Atomic Mass Worksheet and definitely one of these is you, is not it?